Trusted by
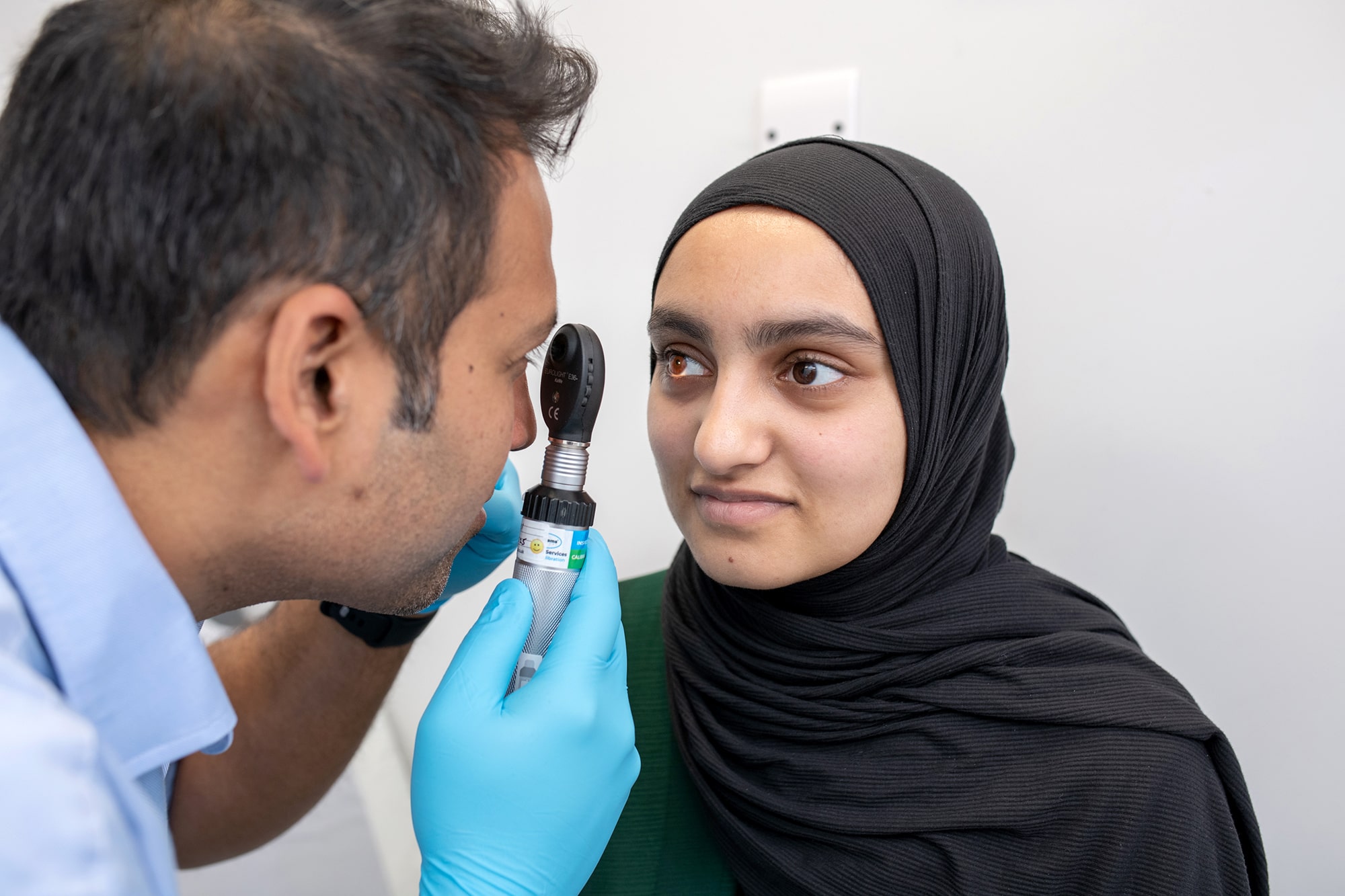
Patients are at the heart of clinical research. By participating in clinical trials, you’re not just accessing cutting-edge healthcare—you’re shaping the future of medicine.
Early access to potential new discoveries – Clinical trials offer an opportunity to access innovative treatments before they become widely available. These treatments are developed with the goal of improving patient care and advancing medical knowledge.
Expert medical care and monitoring – Throughout the trial, you’ll receive regular health check-ups and close monitoring from experienced healthcare professionals. Your well-being is our priority, and our team is dedicated to providing high-quality care and support at every stage.
Contributing to future treatments – By taking part in a clinical trial, you are helping researchers develop better treatments for conditions like yours. Your participation provides valuable insights that can potentially improve care for future patients and contribute to the advancement of medical science.


Find a trial that’s right for you

Alcohol Consumption Clinical Trials
Find our more
C. diff Clinical Trials
Find our more
Fatty Liver Disease Clinical Trials
Find our more
Weight Loss Clinical Trial
Find our more
Pandemic Flu Clinical Trial
Find our more
High / Bad Cholesterol Clinical Trial
Find our more
Menstrual Migraine Clinical Trials
Find our morePanthera by the numbers.
5,000 patients seen in 2024
1st COVID vaccine deployed in the UK
Patient Satisfaction Score of 87%
5,000 patients seen in 2024
1st COVID vaccine deployed in the UK
Patient Satisfaction Score of 87%
Panthera by the numbers.
A snapshot of the data behind our dedication; real results, real reach, real difference. From patient visits to trial success, our numbers tell a story of trust, scale and impact. See how Panthera is redefining clinical research, one number at a time.
Proven performance that delivers: Your trusted partner in clinical research
Faster Start-up
Measurable value is delivered across every stage of your clinical trial. Our streamlined start-up processes consistently reduce time to first patient enrolment, accelerating timelines without cutting corners.
Panthera accelerates clinical trial site start-up by over 3 months compared to the public sector—completing contracting, submissions, and green-light in just 4.5 months vs. 8 months in the NHS.
Increased Retention
By recruiting highly qualified patient volunteers through targeted strategies, we enhance retention and minimise dropouts, ensuring smoother trial progression.
Earlier green-light enables Panthera to begin patient pre-screening and enrolment sooner, improving recruitment momentum and reducing dropout risk caused by long delays.
Return on Investment
Our tailored, efficient approach translates into significant cost savings—often hundreds of thousands annually—without compromising on quality or results.
By shortening site activation timelines, Panthera helps sponsors reduce trial delays and associated costs—saving significant budget annually through more efficient patient onboarding.
Your questions answered
Apply for a trial-
A clinical trial protocol is a comprehensive plan that outlines the study’s objectives, design, methodology, statistical considerations, and organisation. It specifies:
- The purpose of the study
- Eligibility criteria for participants
- Details of tests, procedures, medications, and dosages
- The schedule of activities
- Information to be collected about participants
This protocol ensures the study is conducted consistently and safely across all participants.
-
Clinical trials encompass various study types, including:
- Prevention Trials: Explore methods to prevent diseases in individuals who have not had them or to prevent recurrence.
- Treatment Trials: Test new treatments, drug combinations, or approaches to therapy.
- Diagnostic Trials: Seek better tests or procedures for diagnosing specific conditions.
- Screening Trials: Evaluate ways to detect diseases or health conditions.
- Quality of Life Trials: Aim to improve comfort and life quality for individuals with chronic illnesses.
Many trials compare a new product or therapy to existing treatments or placebos to determine effectiveness. Potential participants are informed beforehand if placebos are part of the study design.
-
Clinical trials progress through several phases, each designed to answer specific research questions:
- Phase 1: Involves a small group (20-80 participants), often healthy volunteers or patients with advanced diseases. The focus is on evaluating safety, identifying side effects, determining safe dosage ranges, and understanding how the body processes the investigational product. This phase typically lasts several months, with about 70% of studies advancing to Phase 2.
- Phase 2: Enrols 100-300 participants with the condition under study to further assess safety and effectiveness. These trials may be randomised and controlled, comparing the investigational product to standard treatments or placebos. Blinding is often used to prevent bias. Phase 2 studies usually take up to two years, with approximately 33% progressing to Phase 3.
- Phase 3: Includes larger groups (thousands of participants) to confirm effectiveness, monitor side effects, and compare the investigational product to standard treatments. These randomised and blinded trials can last from one to four years, with 25-30% successfully leading to regulatory approval submissions.
- Phase 4: Conducted after regulatory approval, these post-marketing studies gather additional information on risks, benefits, and optimal use in various populations over extended periods. Findings can influence whether a product remains on the market or if usage guidelines need adjustments.
-
Participating in a clinical trial offers several potential benefits:
- Access to New Treatments: You may receive investigational therapies not yet available to the public, potentially improving your health condition.
- Contribute to Medical Research: Your involvement aids in understanding how treatments work across different populations, helping to develop better therapies for future patients.
- Comprehensive Care: Participants receive close monitoring and additional healthcare assessments related to the study.
- Voluntary Participation: You can withdraw from the study at any time without any obligation.
Each study has specific eligibility criteria, so not everyone who applies will be accepted.
-
Participation typically involves several stages:
- Pre-Screening: Researchers identify potential participants through databases, advertisements, or health screenings and conduct brief interviews to assess initial eligibility.
- Informed Consent: If you are a potential candidate, you’ll meet with the research team to discuss the study in detail. You’ll receive an informed consent document outlining the study’s purpose, procedures, risks, and benefits. Signing this document indicates your voluntary agreement to participate.
- Screening Visit: After consent, you’ll undergo assessments such as medical history reviews, physical exams, and laboratory tests to confirm eligibility based on the study’s criteria.
- Study Visits (Treatment Visits): Eligible participants attend scheduled visits to receive the investigational product or control, with ongoing monitoring and assessments as per the study protocol.
- End of Study Visit: Upon completing the treatment phase, final evaluations are conducted to gather data on the investigational product’s effects.
- Follow-Up Safety Visit: Some studies require additional visits after treatment concludes to monitor for any lasting effects or side effects.
-
Before deciding whether to take part in a clinical trial, it’s important to have all the information you need. Here are some key questions you might want to ask:
- What is the study aiming to find out? Understanding the purpose of the research can help you decide if it’s right for you.
- What tests and procedures are involved? Ask about the types of tests, how long they take, and what they involve.
- How often will I need to visit the clinic? Knowing the time commitment required can help you plan ahead.
- Will I need to stay in the hospital? If so, find out how often and for how long.
- Are there any costs involved? Ask whether your participation is covered, including any travel expenses or if your insurance is required to cover certain aspects.
- What happens after the study ends? Find out about any follow-up care or ongoing monitoring.
- What are my other treatment options? Understanding how the trial compares to existing treatments can help you make an informed choice.
- What side effects might I experience? Knowing the potential risks and how they compare to standard treatments is important.
- How long does the study last? This will give you a sense of the overall commitment involved.
-
Yes, your privacy is our priority. Any personal health information (PHI) you share with the clinical research team is kept strictly confidential and will not be shared without your permission, except when required by law.
If you choose to submit your details through our website, with your consent, your information will be securely stored in our clinical research database for potential participation in current and future studies. You can request to have your details removed at any time.
Similarly, if you speak with one of our research specialists over the phone, any information you provide will be recorded in the same secure database. This is entirely voluntary, and you can request its removal at any point. Your confidentiality and peace of mind are important to us.
-
No, there is no cost to take part in a clinical trial. All study-related visits, tests, and procedures are provided free of charge.
If you qualify for a study, you may also receive compensation for your time and travel. The details of any compensation, as well as any reasonable expenses that may not be covered by the study, will be explained during the informed consent process. Our team will ensure you have all the information you need before deciding to take part.
-
The length and number of study visits depend on the specific trial. Typically, visits last at least 30 minutes, but some may take a few hours depending on the procedures involved.
Your first visit—known as the screening visit—is usually the longest. This is because it includes the informed consent process, a review of your medical history, collection of lab samples, and study-related assessments or questionnaires.
Before you decide to take part, we’ll provide clear details on how long each visit is expected to take, how often you’ll need to attend, and the overall duration of the study. Your time is valuable, and we aim to make each visit as smooth and efficient as possible.
Find your nearest Panthera Clinic


-
Glasgow Panthera, Health Innovation Hub, 101 Holmfauld Road, Glasgow, G51 4RY
Discover more -
-
-
-
-
-
Keele University Science & Innovation Park | Keele University, IC7, Keele, Staffordshire, UK, ST5 5BG
Panthera & Industry News

Panthera Biopartners Expands European Footprint with Acquisition of Hungary’s Leading Clinical Research Organisation, OEC

Panthera Expands Glasgow Clinical Research Operations with Move to Health Innovation Hub (HiH)

Why the UK is winning back global clinical trials
Ready to take part?
Join a clinical trial and help shape the medicine of tomorrow, today.
Register for a trial



