Pandemic Flu Clinical Trial
Get involved in the Future of Flu Protection

Join a clinical trial testing a new vaccine designed to protect against serious flu strains that could cause a future pandemic
Early access to potential new treatments. Be among the first to benefit from medical advancements designed to improve
Expert medical care and monitoring. Receive ongoing support from leading healthcare professionals throughout the trial, ensuring your well-being at every step.
Making a difference for future patients. Your participation helps shape the future of weight loss medicine, providing insights that lead to better treatments for others.

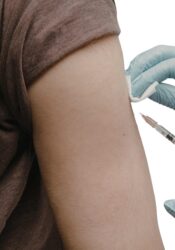

What to expect on your visit
Warm welcome and check in
Our team will will greet you and guide you though the process.
Comfortable waiting area
Relax with seating, refreshments and Wi-Fi whilst you wait.
Meet your specialist
A dedicated professional will explain everything and answer any questions.
Clear instructions
We’ll walk you through each step so you know exactly what to expect
Time for questions
You’ll have the opportunity to ask anything about your participation
Follow-up guidance
We’ll provide details on the next steps and any necessary follow-up visits.
Your questions answered
Apply Now-
A clinical trial protocol is a comprehensive plan that outlines the study’s objectives, design, methodology, statistical considerations, and organisation. It specifies:
- The purpose of the study
- Eligibility criteria for participants
- Details of tests, procedures, medications, and dosages
- The schedule of activities
- Information to be collected about participants
This protocol ensures the study is conducted consistently and safely across all participants.
-
Clinical trials encompass various study types, including:
- Prevention Trials: Explore methods to prevent diseases in individuals who have not had them or to prevent recurrence.
- Treatment Trials: Test new treatments, drug combinations, or approaches to therapy.
- Diagnostic Trials: Seek better tests or procedures for diagnosing specific conditions.
- Screening Trials: Evaluate ways to detect diseases or health conditions.
- Quality of Life Trials: Aim to improve comfort and life quality for individuals with chronic illnesses.
Many trials compare a new product or therapy to existing treatments or placebos to determine effectiveness. Potential participants are informed beforehand if placebos are part of the study design.
-
Participating in a clinical trial offers several potential benefits:
- Access to New Treatments: You may receive investigational therapies not yet available to the public, potentially improving your health condition.
- Contribute to Medical Research: Your involvement aids in understanding how treatments work across different populations, helping to develop better therapies for future patients.
- Comprehensive Care: Participants receive close monitoring and additional healthcare assessments related to the study.
- Voluntary Participation: You can withdraw from the study at any time without any obligation.
Each study has specific eligibility criteria, so not everyone who applies will be accepted.
-
Clinical trials progress through several phases, each designed to answer specific research questions:
- Phase 1: Involves a small group (20-80 participants), often healthy volunteers or patients with advanced diseases. The focus is on evaluating safety, identifying side effects, determining safe dosage ranges, and understanding how the body processes the investigational product. This phase typically lasts several months, with about 70% of studies advancing to Phase 2.
- Phase 2: Enrols 100-300 participants with the condition under study to further assess safety and effectiveness. These trials may be randomised and controlled, comparing the investigational product to standard treatments or placebos. Blinding is often used to prevent bias. Phase 2 studies usually take up to two years, with approximately 33% progressing to Phase 3.
- Phase 3: Includes larger groups (thousands of participants) to confirm effectiveness, monitor side effects, and compare the investigational product to standard treatments. These randomised and blinded trials can last from one to four years, with 25-30% successfully leading to regulatory approval submissions.
- Phase 4: Conducted after regulatory approval, these post-marketing studies gather additional information on risks, benefits, and optimal use in various populations over extended periods. Findings can influence whether a product remains on the market or if usage guidelines need adjustments.
-
A clinical trial is a medical research study involving volunteers to explore new ways to prevent, detect, or treat diseases, illnesses or ailments. These studies assess investigational treatments, which may include new drugs, combinations of drugs, new delivery methods for existing medications, or medical devices. Before approval, these treatments undergo rigorous testing to ensure they are safe and effective. Each trial follows a detailed protocol outlining the study’s purpose, participant criteria, procedures, and duration. A principal investigator, often a medical doctor, leads the research team, which includes various healthcare professionals. Participation is entirely voluntary, and diverse involvement is crucial to understand different responses to the investigational product.
Trial details overview
What’s this clinical trial about?
Flu viruses are constantly changing. Some strains like H5N1 have the potential to spread widely and cause serious illness.
Panthera is conducting a clinical trial to help develop a new investigational vaccine aimed at protecting people against flu strains that could lead to a pandemic. By volunteering, you can play an important role in advancing future public health protections.
Why is this study important?
- Help researchers prepare for future flu pandemics
- Contribute to vaccine science that may protect millions
- Receive trial-related care at no cost
- Compensation may be available for time and travel (where applicable)
Who Can Take Part?
We are enrolling adults in two age groups:
- 18 to 64 years
- 65 years and older
What does participation involve?
- You will receive either the investigational flu vaccine or a placebo
- There is a 75% chance you will receive the investigational vaccine
- Clinical trial visits, health checks, and follow-up will be clearly explained before you decide
- All participants are monitored closely by experienced medical professionals
Who May Not Be Eligible?
You may not be able to take part if you:
- Are pregnant or breastfeeding, or unable to follow contraceptive requirements
- Have had prior or planned surgical treatment for obesity
- Have experienced significant recent weight changes
- Are currently participating in another weight-loss program or using weight-loss medications
- Are currently using or have recently used injectable treatments for diabetes or weight loss
- Have certain medical conditions or lab results that may affect study participation
- Are currently participating in another clinical trial.
- Have a history of pancreatitis
A full eligibility review will be completed during screening
Your Safety Comes First
Your participation is completely voluntary. You can ask questions at any time and may withdraw from the study at any point. All research is conducted under strict safety and ethical guidelines.
In the news

Panthera Joins the Association of Multisite Research Corporations (AMRC)

Panthera’s Success Featured in The Times article about UK life sciences industry

Panthera strengthens commercial team with appointment of two former NIHR executives
Available Panthera Locations

